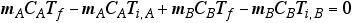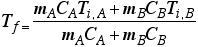Conservation in energy transfer
When energy is lost by a substance it is gained by another, and vice versa.
- This can be shown and measured by the act of dropping a warmer substance into a cooler one, and measuring the respective changes in temperature.
- The net change of energy in the system will be zero.
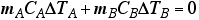
When the two substances are put together, heat will flow out of the warmer one into the cooler one until thermal equilibrium is reached.
- Let the temperature at thermal equilibrium = Tf
- This will be the final temperatures of BOTH substances!
- To find that temperature at which equilibrium is reached,
Expand DT to Tf -Ti for each substance
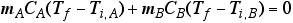 and
and
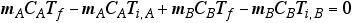
Solve the equation for Tf
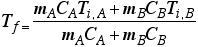
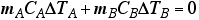
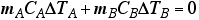
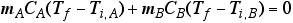 and
and