ERHS PHYSICS
Decay Chain of Uranium Exercise
(online version)
NAME____________________________
Date____________________Per________
Problem: How can a decay chain be shown in a diagram?
Information:
As radioactive elements emit alpha and beta particles, the elements change into a series of other elements.
- Each time there is an ALPHA decay, the mass number is decreased by 4 and the atomic number is decreased by 2. This is because an alpha particle consists of 2 protons and 2 neutrons. The total mass number of such a particle is 4 Atomic Mass Units (2 AMU from the protons, and 2 AMU from the neutrons).
- Each time there is a BETA decay, the mass number remains constant, but the atomic number increases by 1. This is because in order for there to be a beta decay, a neutron in the nucleus must divide into an electron and a proton. The electron (beta particle) is ejected. Its mass is so small that the mass number is unaffected, but there is now one more proton in the nucleus than before.
Purpose: In this investigation, you will learn how to describe the decay chain of uranium-238 by means of a diagram, so that the changes of elements will be easy to see and understand. You will learn what happens to an element during alpha and beta decays, and you will gain further understanding of the periodic table and graphing.
Procedure: All the data you need in this investigation are provided in the decay-chain table you have on this page. You will put the data into a graph-like diagram.
- Construct a graph on a piece of 8.5 x 14 inch paper, with a grid of 11 lines X 33 lines. Plot mass number (independent variable) across the horizontal, or "X" axis, starting with 238 and going down to 206. Look at the example below.
- Plot atomic numbers (dependent variable) along the vertical, or "Y" axis starting at the top with 92 and going down to 82. Again, the first few are shown for you.
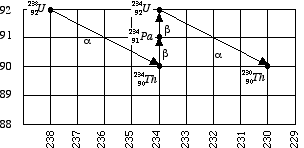
- The first four steps of the decay chain are shown in figure 1 below, to show you how to plot the progress of the decay chain. Notice that the symbol for the emitted particle is written next to the arrow.
- The first step in the decay of U-238, an alpha decay, produces a new atom with an atomic number two less than the original one, and a mass number four less than the original one. All alpha decays follow this pattern, since the alpha particle carries away two protons and two neutrons.
- The second step is a beta decay. The new atom has an atomic number one greater than the original atom. The mass number is unchanged, All beta decays follow this pattern, since the beta particle carries away one negative charge and practically no mass. The third step is another beta decay, and the fourth is an alpha decay.
- Duplicate the first four steps as shown on your graph. Then, using information from the table below, complete the diagram of the decay chain, using exactly the same pattern for labeling as shown.
- Write a nuclear equation for each step indicating the type of decay and all resulting particles.
The first 4 steps of the decay chain are shown above. Compare these steps to those listed in the table on the back of this page. You are to complete the graph, INCLUDING the steps shown in figure 1 above. Label each data point as shown, and indicate whether the step represents alpha or beta decay. Just follow the model and you'll be doing the right thing.
|
|
|
Decay Chain of Uranium |
|
|
| Step |
element
|
symbol
|
event
|
new element
|
symbol
|
| 1 |
Uranium-238 |
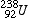 |
alpha-decays, forming |
Thorium-234 |
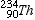 |
| 2 |
Thorium-234 |
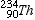 |
beta-decays, forming |
Protactinium-234 |
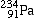 |
| 3 |
Protactinium-234 |
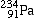 |
beta-decays, forming |
Uranium-234 |
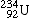 |
| 4 |
Uranium-234 |
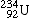 |
alpha-decays, forming |
Thorium-230 |
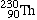 |
| 5 |
Thorium-230 |
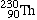 |
alpha-decays, forming |
Radium-226 |
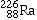 |
| 6 |
Radium-236 |
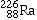 |
alpha-decays, forming |
Radon-222 |
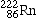 |
| 7 |
Radon-222 |
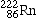 |
alpha-decays, forming |
Polonium-218 |
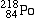 |
| 8 |
Polonium-218 |
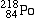 |
alpha-decays, forming |
Lead-214 |
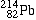 |
| 9 |
Lead-214 |
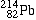 |
beta-decays, forming |
Bismuth-214 |
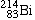 |
| 10 |
Bismuth-214 |
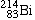 |
beta-decays, forming |
Polonium-214 |
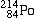 |
| 11 |
Polonium-214 |
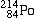 |
alpha-decays, forming |
Lead-210 |
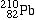 |
| 12 |
Lead-210 |
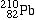 |
beta-decays, forming |
Bismuth-210 |
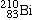 |
| 13 |
Bismuth-210 |
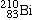 |
beta-decays, forming |
Polonium-210 |
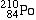 |
| 14 |
Polonium-210 |
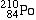 |
alpha-decays, forming |
Lead-206 |
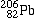 |

