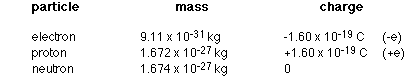Background information:
All matter is made up of atoms.
- In turn, all atoms are made up of particles called protons, neutrons, and electrons.
- Protons are positively charged, neutrons are neutral, and electrons are negatively charged.
- Protons and neutrons make up the center of an atom, and the electrons orbit around them.
- Electrons are free to move, and can move from one atom to another.
Electrons can be passed from the atoms of one object to another by rubbing the objects together.
- When the atoms that make up an object have an abundance of electrons, the object is said to be negatively charged. When the object loses electrons, it is said to be positively charged.
Most of the time, electrons between objects are in balance, and the objects are neutral. Some objects tend to lose electrons easily, while others tend to gain them more easily.
- Wool is a material which will give up electrons rather easily, and plastic will gain these lost electrons.
- Therefore, when we rub the wool against the plastic, the plastic will have a negative charge.
- This negative charge will attract both positively and neutrally charged objects to it, so that it can share the extra electrons, and make everything neutral.
- The goal of things in nature is for everything to be neutral.
- Read about the lightning research at Langmuir Laboratory in New Mexico
Read this website and answer the 4 questions:
http://www.glenbrook.k12.il.us/gbssci/phys/class/estatics/u8l2a.html
Static electricity is the imbalance of positive and negative charges. Various machines have been produced over the years that will produce static charge. If you like to build stuff, check out one or both of these pages on the internet:
Electrostatic machines
Static Generators
We will be demonstrating the Van de Graaff machine in class. Here is a web page made by ERHS 9th graders about their experiences with the machine.
Conductors and Insulators
Materials are divided into two basic groups: conductors and insulators.
- Within a conductor, electrons move freely throughout the entire substance. Therefore, when an ungrounded conductor becomes charged, the entire volume of the conductive body assumes a charge of the same voltage and polarity. A charged conductor can be neutralized by connecting it to earth ground.
- Charge of one polarity can remain on a conductor as long as it is isolated from ground.
- A charged insulator can remain charged for many hours. Opposite polarity charges can exist on an insulator at the same time.
- Charges will not migrate on insulators.
- Grounding insulators neither removes nor prevents surface charges, therefore, other means must be used for neutralizing static on insulators.
- Within an insulator, the flow of electrons is very limited.
- Because of this, an insulator may retain several static charges of different polarities and potentials at various areas on its surface. This accounts for why certain areas of a material may stick together and others may repel each other.
Establishing an electric charge on an object
Charging by induction
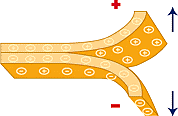
- When you bring a negatively charged object close to another object, electrons in the second object will be repelled from the first object and congregate more toward the other end. Therefore, that end will have a negative charge. This process is called charging by induction. This leaves local excesses of positive and negative charges on the second object.. the round object in the diagram below.

- A highly charged object is surrounded by a static charge field. If an isolated or ungrounded conductive object enters into this static field, it too will become charged. This creates the possibility of electrostatic discharge to some other conductive object, which could result in an arc of sufficient energy to ignite combustibles or destroy sensitive electronic components.
Charging by conduction
- When a negatively charged object touches a neutral body, electrons will spread on both objects and make both objects negatively charged. This process is called charging by conduction, or triboelectrification. The other case, positively charged object touching the neutral body, is just the same in principle.
- When substances become charged by triboelectrification, electrons migrate from the surface of one material to the surface of the other. Upon separation of the two surfaces, one surface loses electrons and becomes positively charged. The other surface gains electrons and becomes negatively charged.
- Scientists have ranked materials in order of their ability to hold or give up electrons. This ranking is called the triboelectric series. A list of some common materials is shown here. Under ideal conditions, if two materials are rubbed together, the one higher on the list should give up electrons and become positively charged. You can experiment with things on this list for yourself
|
Triboelectric Series
|
|
your hand
|
|
glass
|
|
your hair
|
|
nylon
|
|
wool
|
|
fur
|
|
silk
|
|
paper
|
|
cotton
|
|
hard rubber
|
|
polyester
|
|
polyvinylchloride plastic
|
homework: set#1: p 477 questions 1-8 set#2: problems 20-28
Static electricity lab: Control-click this link and open the activity page in a new window.
video: Understanding electricity
Read about the tethered satellite experiment you saw in the video
You may also be interested in this company's work:
Haverfield Corporation (AgRotors, as seen in the video, was absorbed by Haverfield.)
Read more about the Utah Arm you saw in the video.
Highly Recommended internet resource for this chapter:
http://library.advanced.org/10796/ch11/ch11.htm
|
|